The Challenge: Supply Chain Resilience vs. Quality Compliance
For ASEAN-based pharmaceutical manufacturers, diversifying the supplier base for Active Pharmaceutical Ingredients (APIs) and Key Starting Materials (KSMs) is no longer optional—it is a strategic necessity to mitigate geopolitical risks and optimize COGS. However, diversification often introduces variability. The core challenge lies in maintaining stringent Good Manufacturing Practice (GMP) standards and ensuring that cost-effective sourcing does not lead to batch failures, regulatory “Warning Letters,” or compromised Service Level Agreements (SLAs) asall these outweigh the cost savings and source diversity. So, can we get the best of both worlds?
The Solution: A Technical Framework for Embedded Quality Assurance
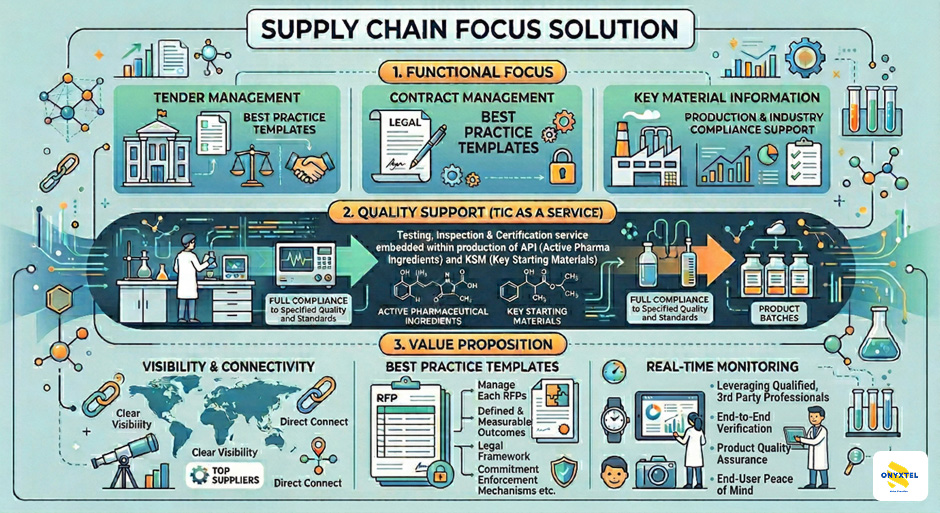
We provide a comprehensive technical solution that moves beyond traditional procurement, embedding Testing, Inspection, and Certification (TIC) directly into the supplier’s production lifecycle. Our approach is built on four technical pillars:
1. Lifecycle Visibility & Material Characterization
We conduct exhaustive audits of the supplier landscape, focusing on “next-level” material visibility. By analyzing the synthesis pathways of APIs and the impurity profiles of KSMs, we ensure that new suppliers align with the manufacturer’s established Marketing Authorization (MA) and technical dossiers.
2. Outcome-based Framework with focus on Risk Prevention
We deploy best-practice, template-driven RFP/RFx and contract management frameworks. These are not merely commercial documents; they are technical specifications that define critical quality attributes (CQAs) and critical process parameters (CPPs), ensuring that price negotiations never supersede technical requirements and quality & availability remain core considerations.
3. Operationalized TIC Integration
Rather than relying on “Quality at Entry” (testing upon arrival), we operationalize TIC mechanisms at the source. By embedding third-party certification into the supplier’s batch release process, we ensure a “Right First Time” (RFT) ratio. Shipments are only initiated upon the digital verification of the Certificate of Analysis (CoA) and compliance with pre-defined quality gates.
4. ESG & Regulatory Data Integration
To support the evolving regulatory landscape, our solution captures secondary data points, including the Supply Chain Carbon Footprint and environmental impact metrics. This provides a turnkey solution for ESG submissions and local pharmaceutical standard compliance, ensuring the supply chain is as sustainable as it is clinically compliance.
The Outcome
This holistic offering empowers manufacturers to achieve a diversified, high-velocity supply chain. By bridging the gap between procurement and the production delivery, we construct a quality assurance mechanism that competitive pricing and supply security coexist with uncompromised product integrity.